"Throughout my professional career of serving the MRI community for more than 30 years, I have been committed to ensuring the safety of patients and healthcare workers."
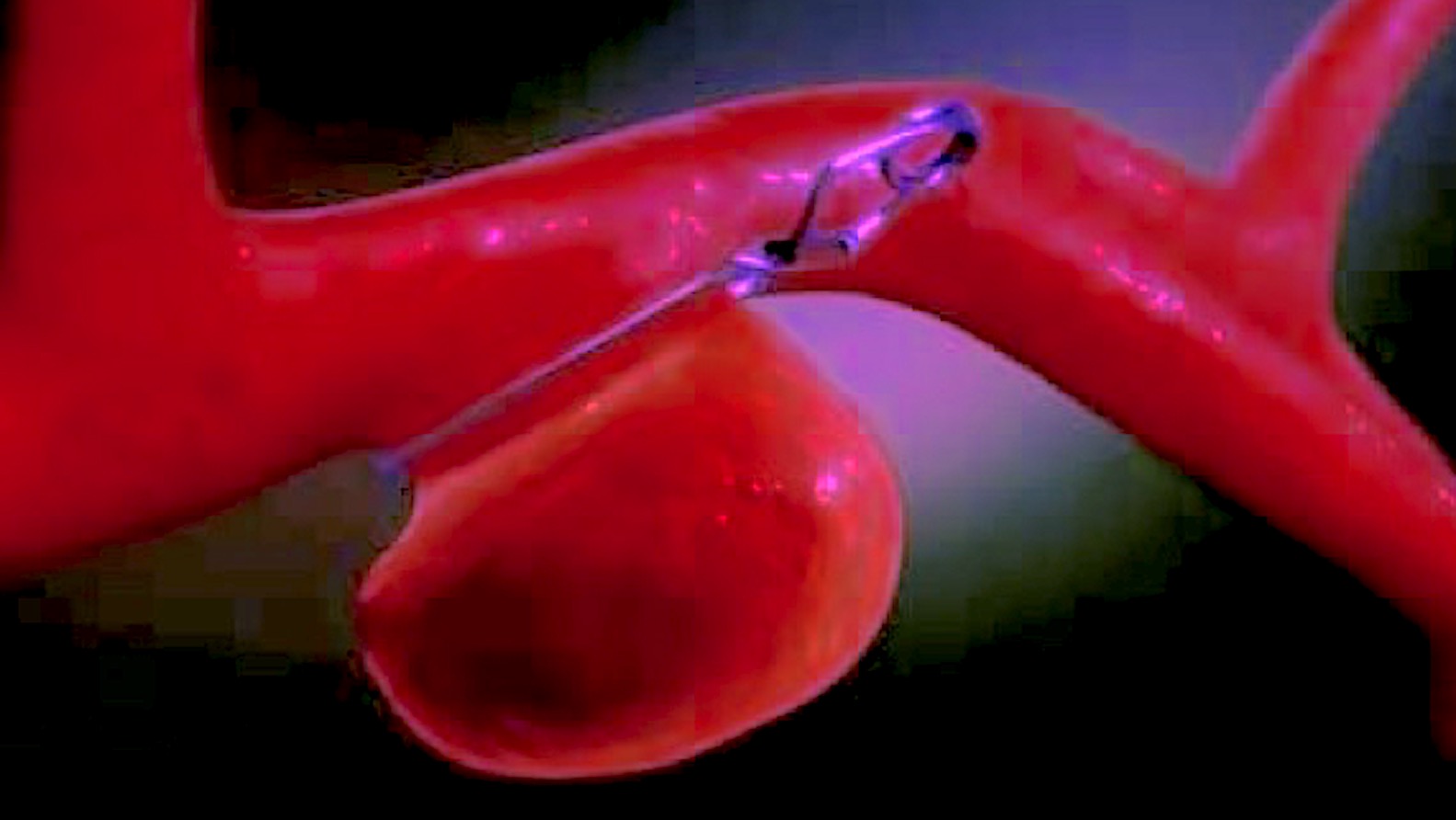
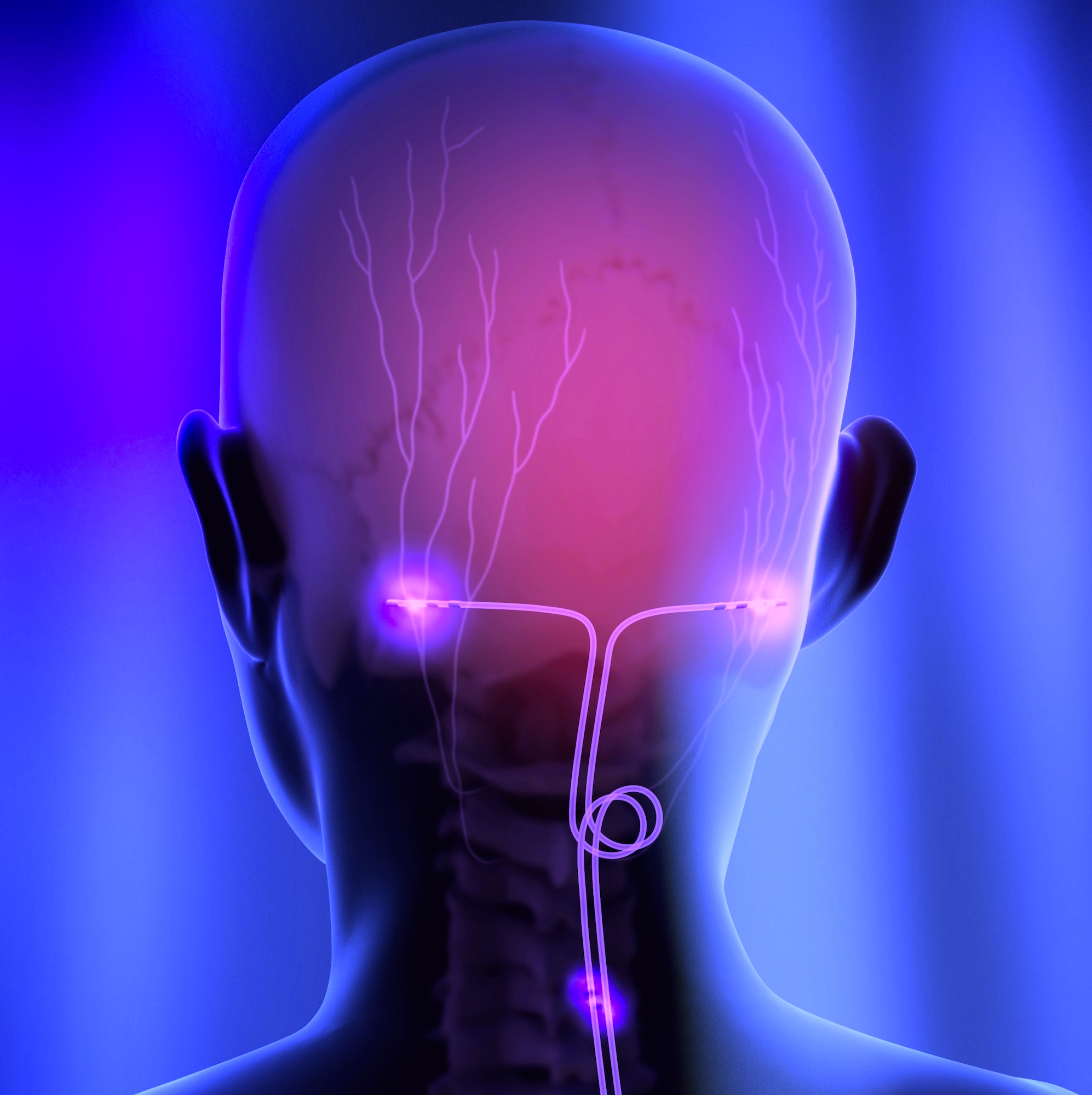
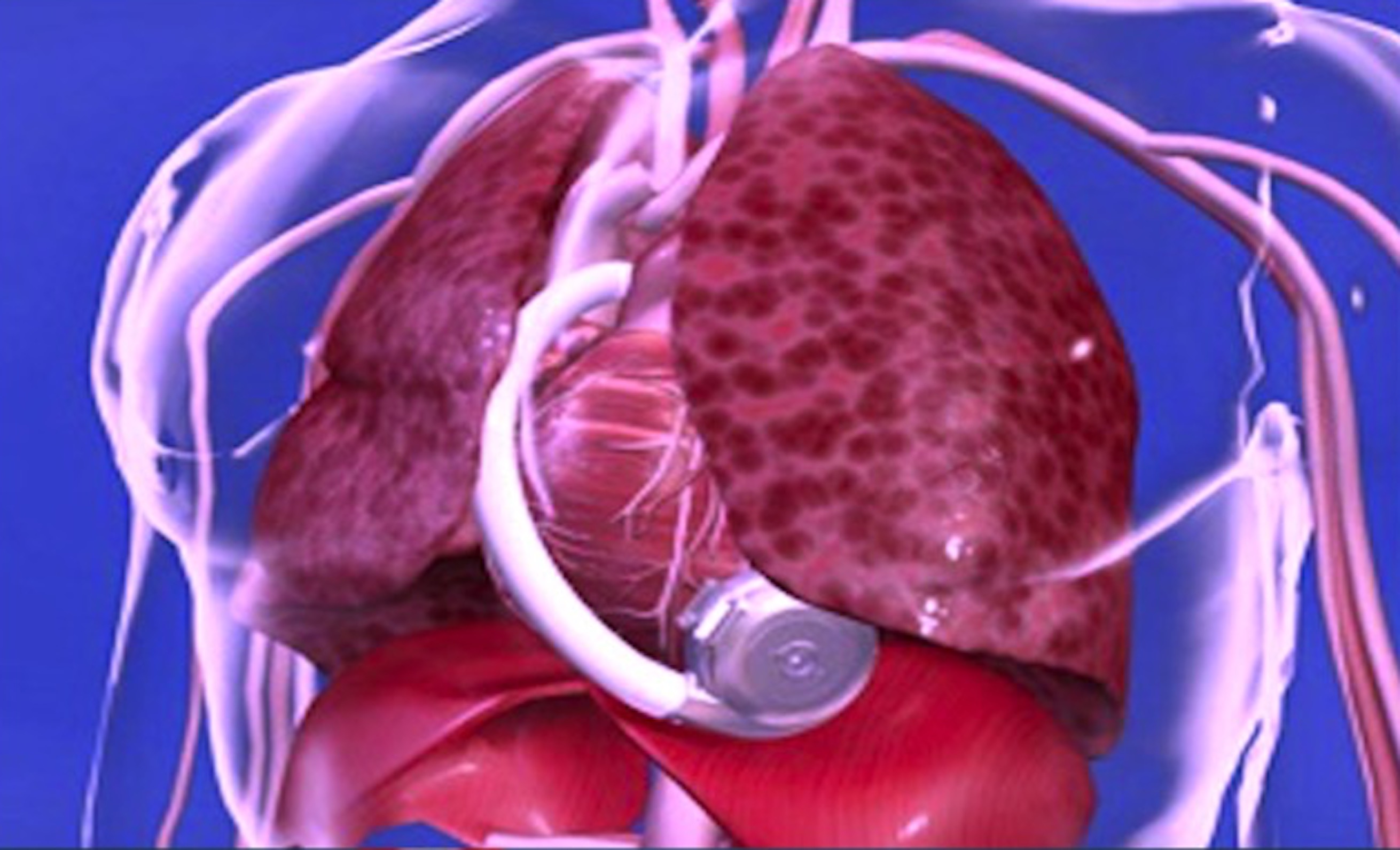
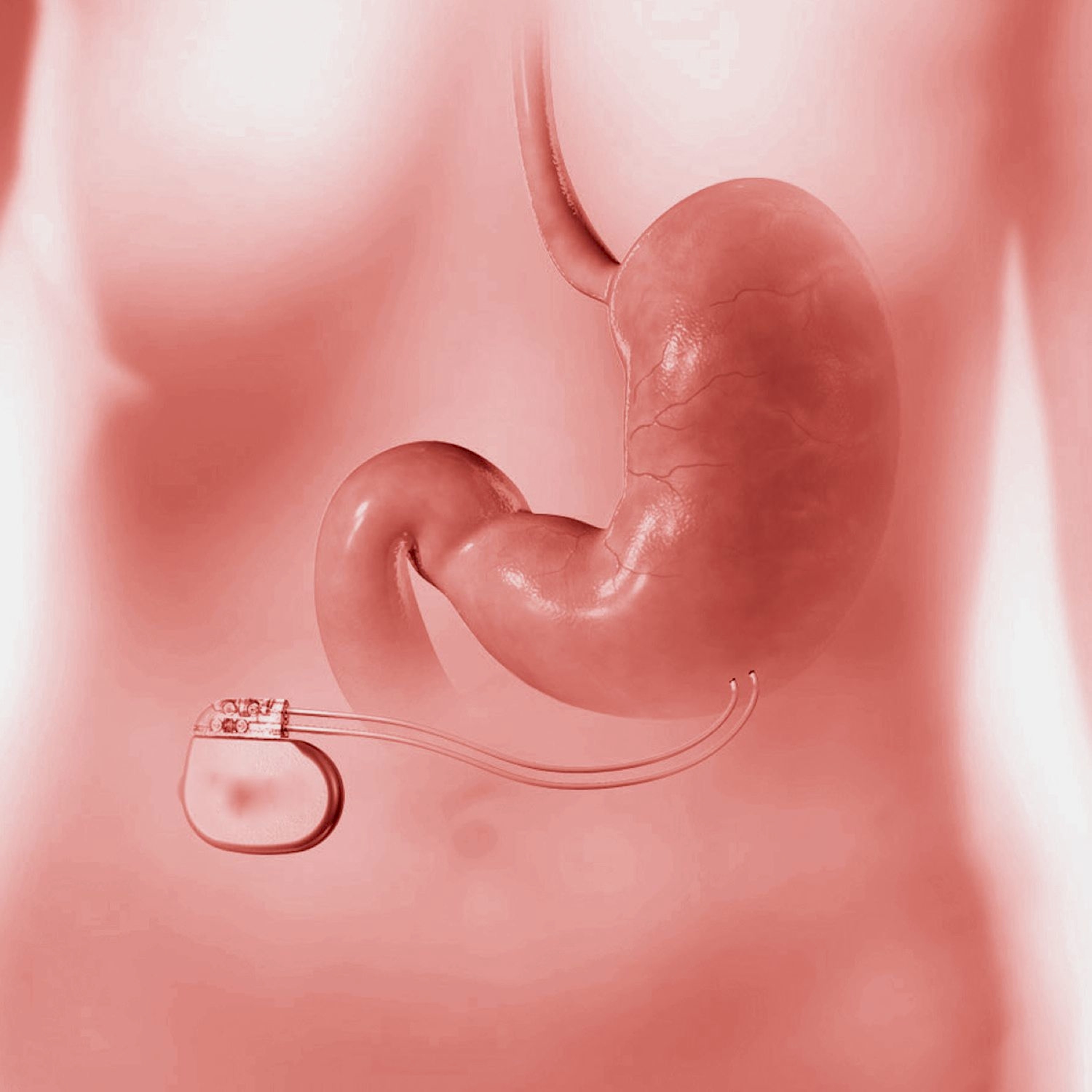
Shellock R&D Services, Inc. is the World’s most experienced MR-safety testing company that conducts comprehensive evaluations of implants, devices, objects, and materials.
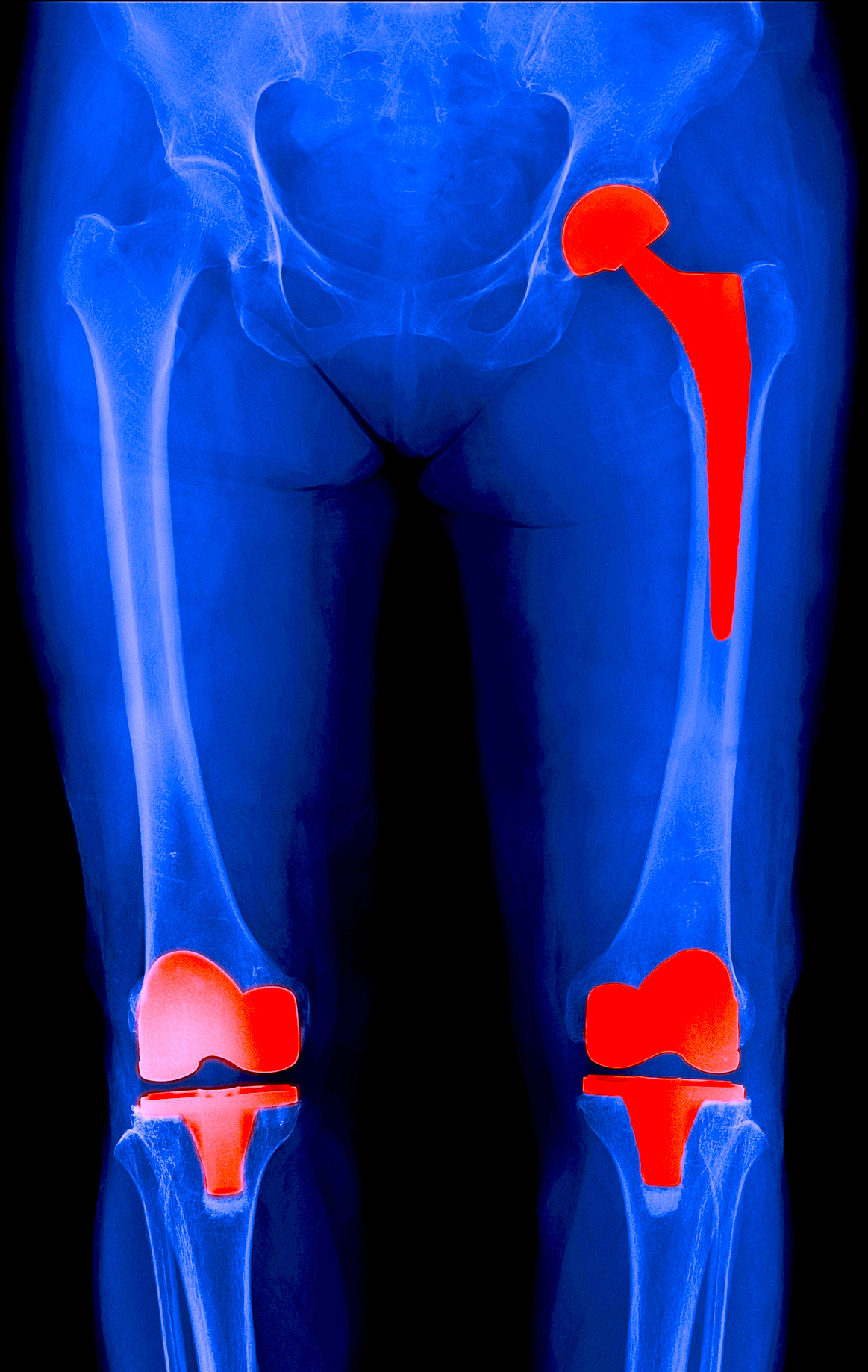
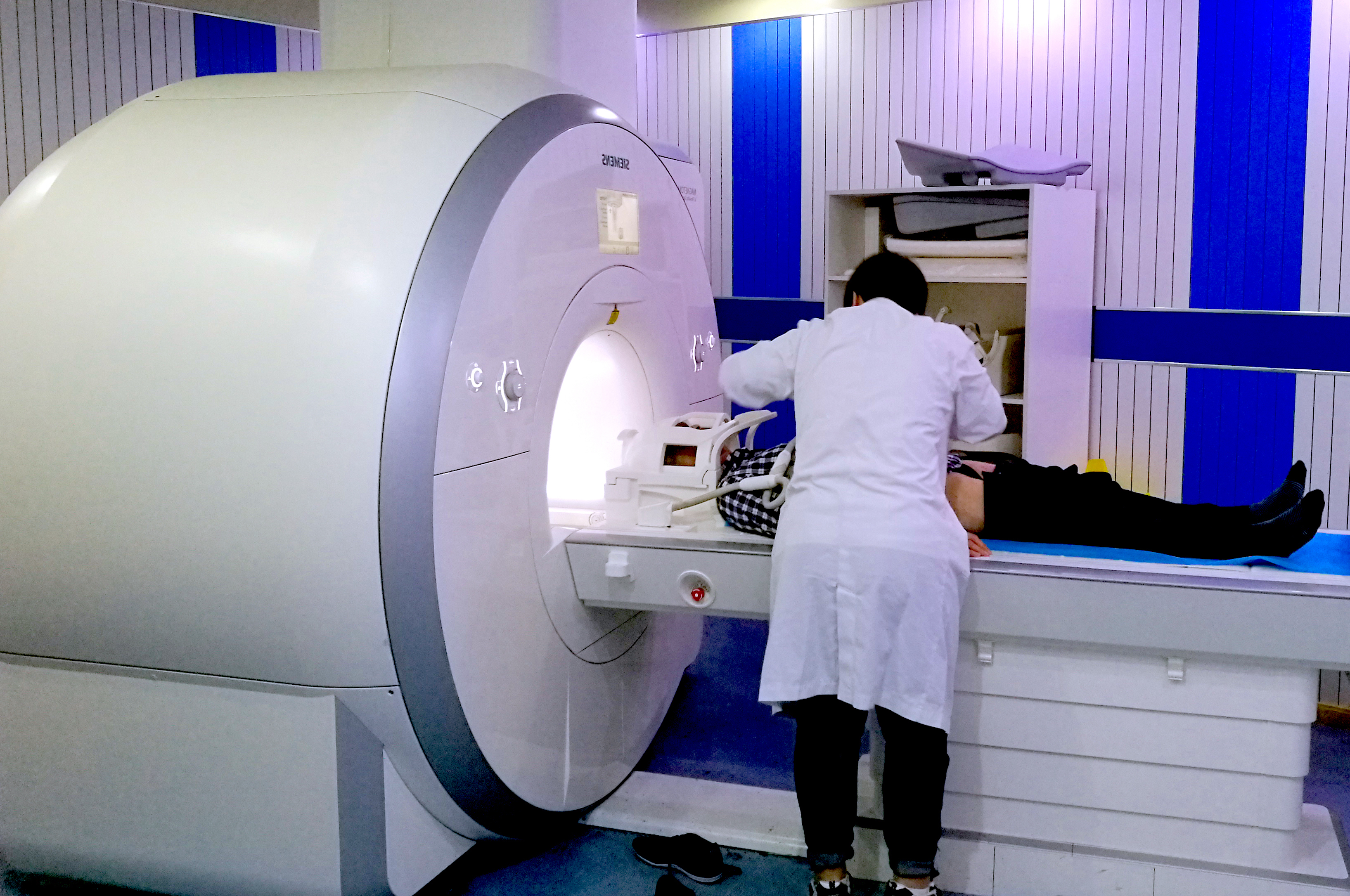
Shellock R&D Services, Inc. has access to MR systems operating from 0.2-Tesla to 7.0-Tesla, including dedicated-extremity and interventional MR systems. Testing includes assessment of magnetic field interactions, heating, induced currents, and artifacts.
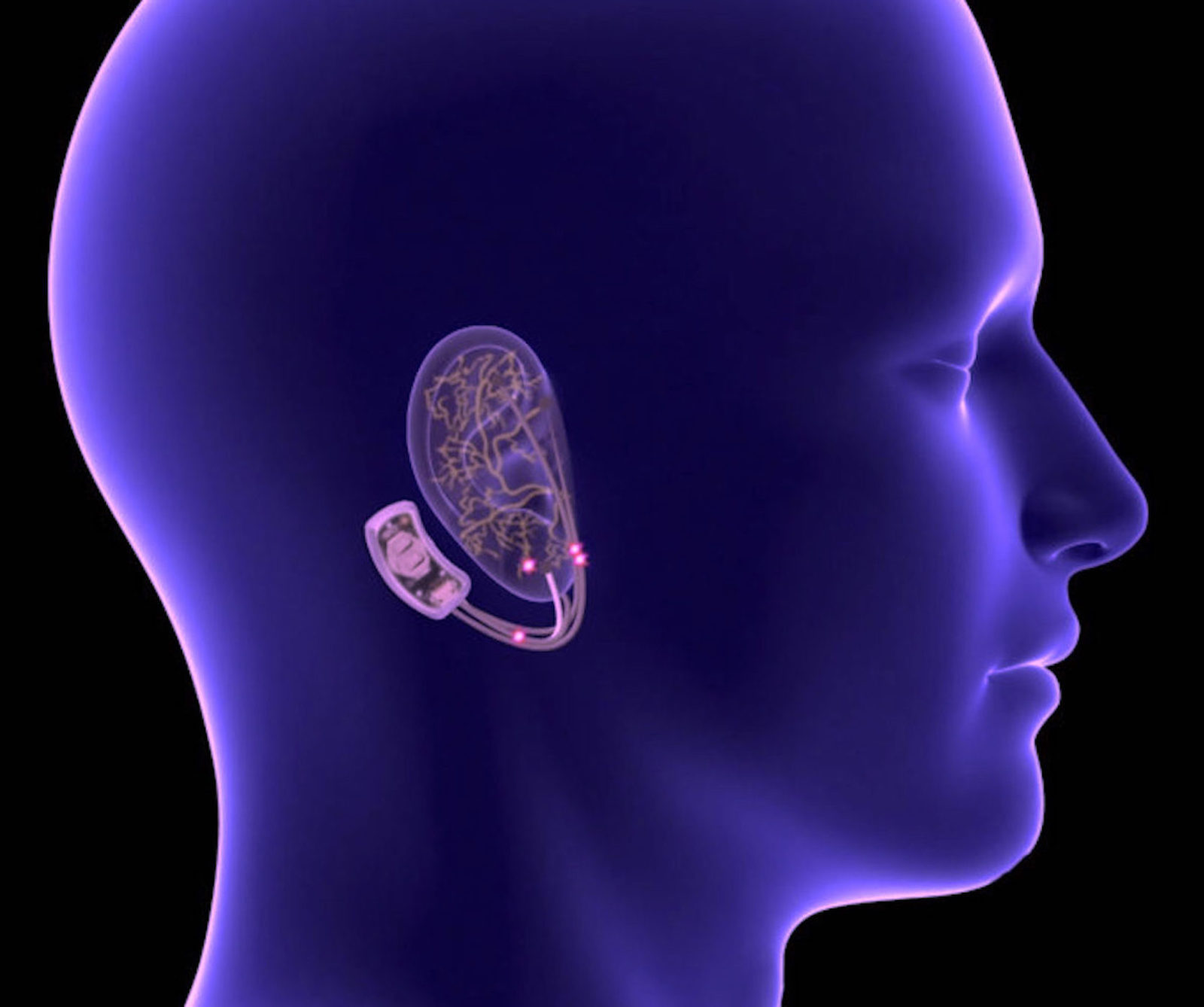
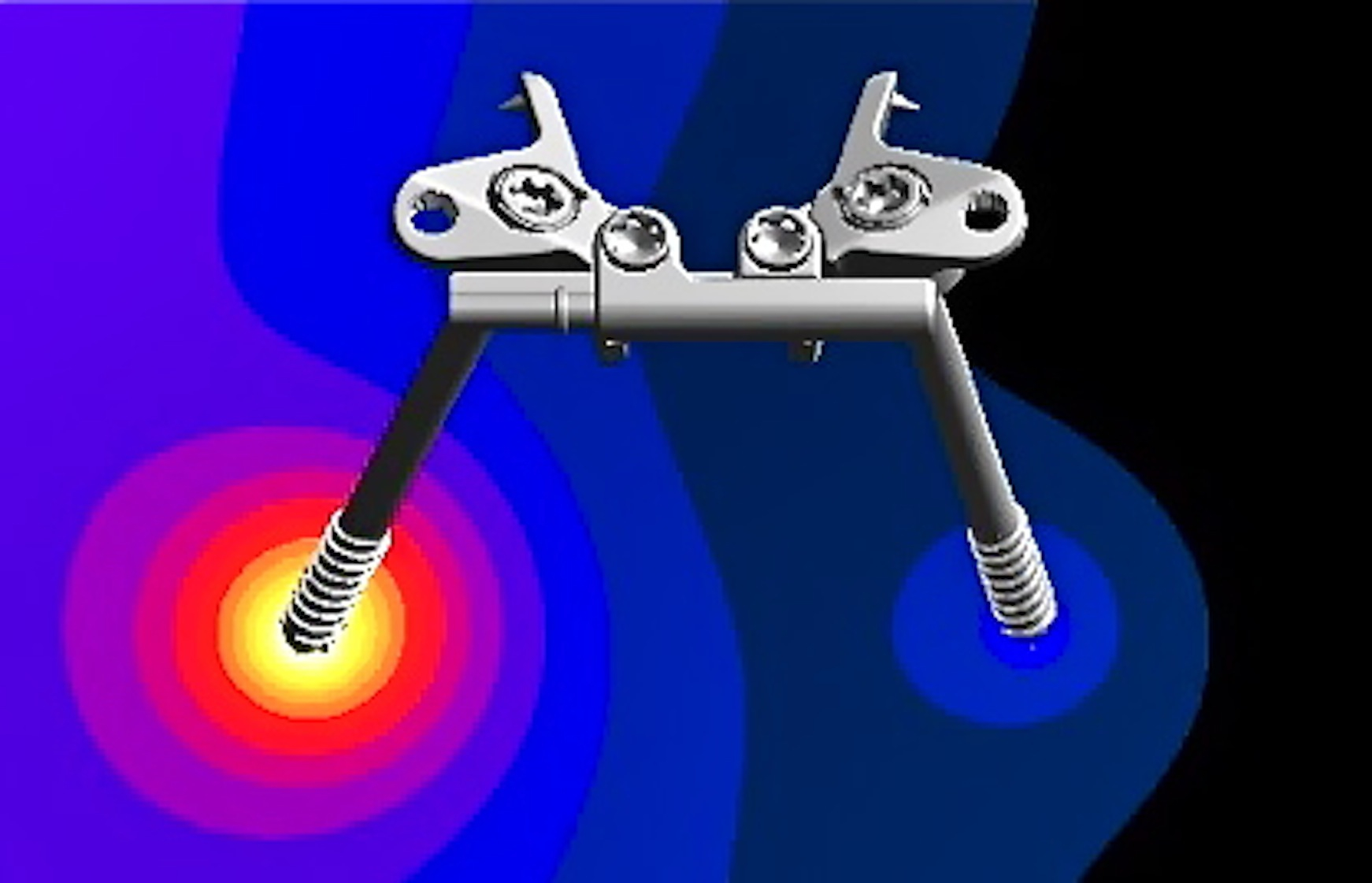
Conducting MRI safety assessments of implants with complicated shapes or multiple sizes require computer simulations. In cooperation with our professional partner Dr. Wolfgang Kainz from HPC for MRI Safety, we offer this vital service. For more information, visit: https://www.hpcmris.com/
Shellock R&D Services, Inc. has access to MR systems operating from 0.2-Tesla to 7.0-Tesla, including dedicated-extremity and interventional MR systems. Testing includes assessment of magnetic field interactions, heating, induced currents, and artifacts.
Conducting MRI safety assessments of implants with complicated shapes or multiple sizes require computer simulations. In cooperation with our professional partner Dr. Wolfgang Kainz from HPC for MRI Safety, we offer this vital service. For more information, visit: https://www.hpcmris.com/
- Frank G. Shellock, Ph.D., President, Shellock R&D Services Inc., (dba, Magnetic Resonance Safety Testing Services)